How Soap perform cleansing action?
Soaps and detergents perform the cleaning action because the polar parts of their molecules are water soluble due to hydrogen-bonding and non-polar parts remain outside water.

Soaps and detergents perform the cleaning action because the polar parts of their molecules are water soluble due to hydrogen-bonding and non-polar parts remain outside water.

Solids can be classified on the basis of the regular arrangements of constituent atoms, ions or molecules. There are two types of solids in this respect: Crystalline Solids Those solids in which atoms, ions or molecules are arranged in a definite three-dimensional patterns with regular and repetitive characteristics are called crystalline solids. Amorphous Solids The…
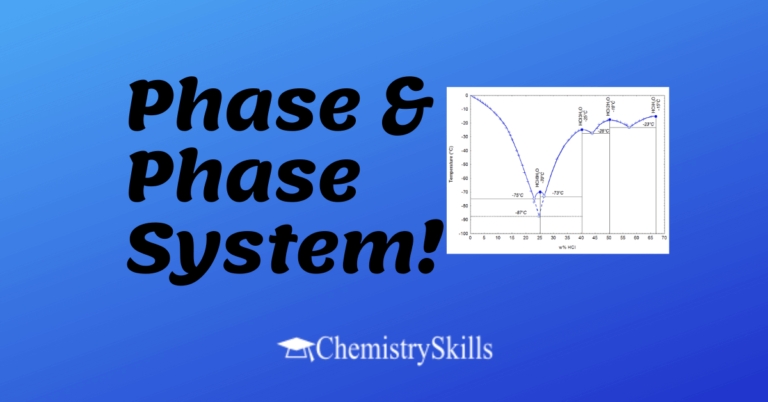
Phase Matter with uniform properties and a fixed composition is called a phase. Types of Phase System There are two types of phase. (i) Homogeneous Phase System (Single Phase System) Water at normal pressure and at room temperature exists as a single liquid phase, i.e. all the properties of water are uniform throughout this liquid…

Whenever matter undergoes a physical or’ a chemical change, it is always accompanied by an energy change in the form of heat. This change in energy is the quantitative measure of the difference in the strength of intermolecular forces. In case of physical change at constant pressure called enthalpy change. Enthalpy change is of three…

A technique which is used to find out relative abundance of isotopes with respect to their atomic masses in the form of graph, is called Mass Spectrometry. Is mass spectrometry quantitative or qualitative? Mass spectrometry is used for quantitative as well as qualitative analysis. What is a mass spectrometer used for? An instrument, which…

In 1850, a French mathematician August Bravis observed that crystal lattice of substances can be categorized into seven types. These are called Bravis Lattices and their units cells are referred to as Bravis unit cells. A crystal system may be identified by the dimensions of a unit cell along its three edges or axe, a,…

Distinct state of matter, containing significant number of electrically charged particles to affect its electrical properties and behavior is called PLASMA. Plasma Formation: When more heat is applied, the atoms or molecules may be ionized. The atom loses one electron and develops a net positive charge. It becomes an ion. When gas is heated…