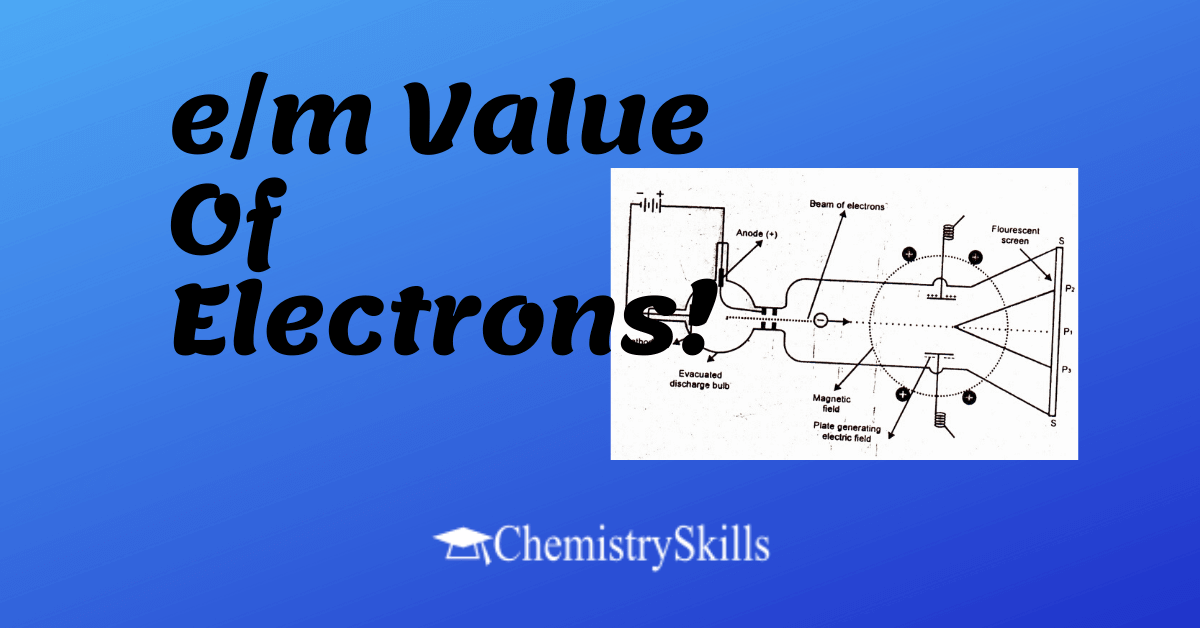
Measurement of Charge to Mass ratio of Electron
i) In order to measure the e/m value of electrons J.J Thomson in 1897 devised an instrument consisting of a discharge tube.
ii) The cathode rays are allowed to pass through electric and magnetic fields.
iii) When both the fields are off, the beam of cathode rays consisting of electrons produce bright luminous spot at P1 on the fluorescent screen.
iv) The electric field being in the plane of the paper of diagram. The north and south poles of magnetic field are perpendicular to the plane of the paper.
v) When only magnetic field is applied the cathode rays are deflected in a circular path and full at the point P3.
vi) When only electric field is applied cathode rays produce a spot at P2.
vii) Both electric and magnetic fields are then applied simultaneously and their strengths adjusted in such a way their cathode rays again hit the point P1.
viii) In this way by comparing the strengths of the two fields one can determine the e/m value of electrons. It comes out to be 1.7588 x 1011 coulombs per kg.