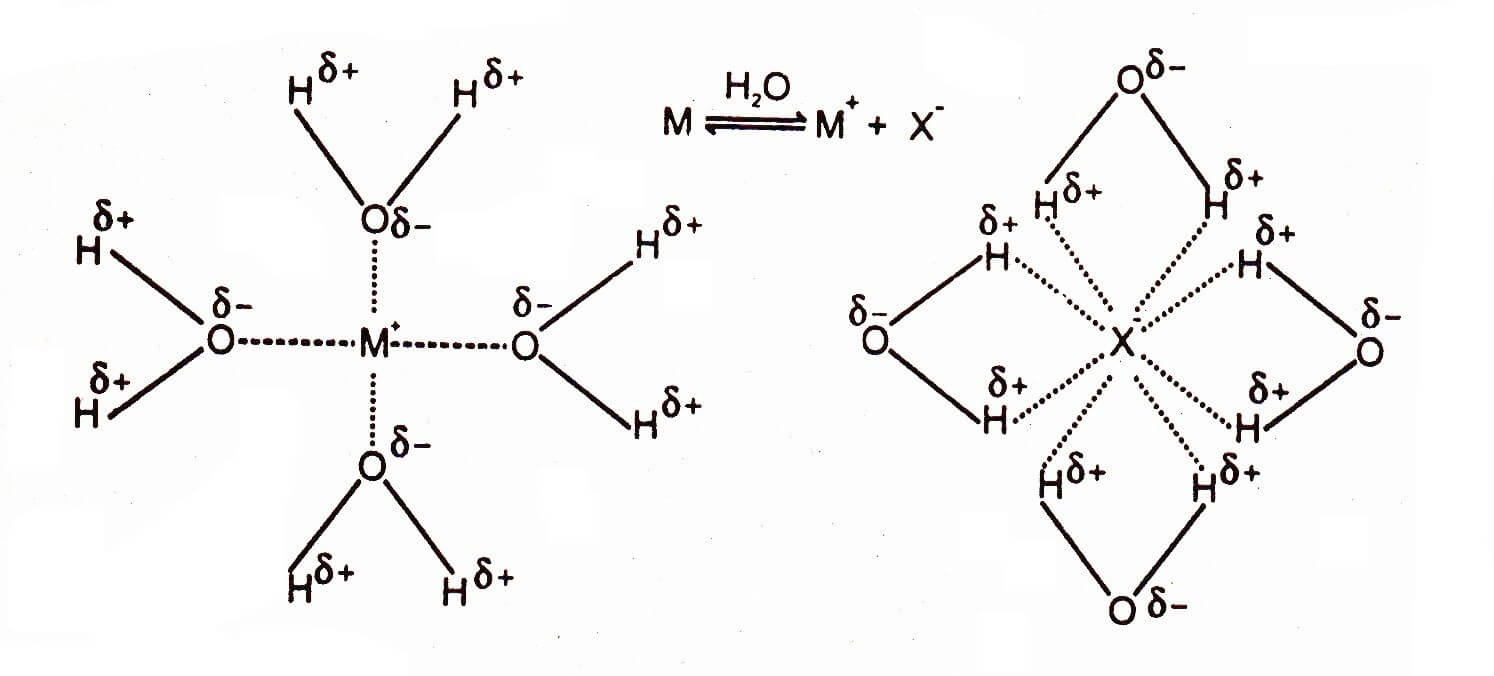
Ion-Dipole Intermolecular Forces
Definition:
The forces of attraction between ions and the molecule of the polar solvent are called ion-dipole forces.
Explanation:
Molecules with ionic bonds are soluble in polar solvents. Solvent molecules break the lattice of ionic compounds and ions are set free. Oppositely charged ions are produced by ionic compounds in polar solvents which surrounded by opposite poles of polar solvents.. The negative ends of the dipole of water are attracted towards the cation (M) while the positive ends towards the anion (X). That’s why ionic compounds are soluble in water. The forces of attraction between ions and polar solvents are called ion-dipole forces.