Crystal Structure of Diamond | Properties
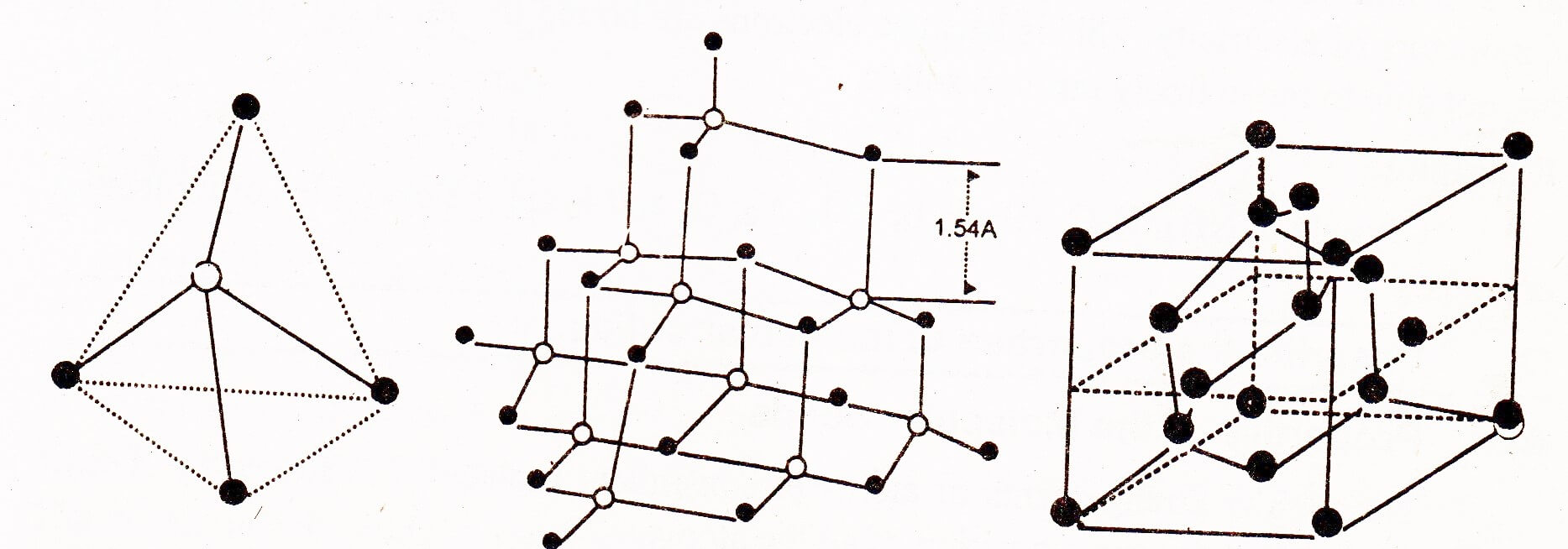
Diamond is crystalline (Allotropy) carbon. In the outermost shell of carbon, which is four, the atomic orbitals (1s and 3p) undergo sp3 hybridization to give four sp3 hybridized orbitals which are directed in space along the four corners of a tetrahedron.
This is the unit cell of diamond and a large number of such unit cells undergo sp3 – sp3 overlapping to form a huge structure. Every carbon atom in diamond is attached with 4 more carbon atoms. The bonds between carbon atoms are covalent which run through the crystal in three dimensions. All the bond angles are 109.50 and the bond lengths are 154 pm. The whole lattice is, therefore, continuous and because of the continuity of C-C covalent bonding the entire diamond crystal behaves as a huge or giant three-dimensional carbon molecule. This is also called a macro-molecule. The overall structure of diamond looks face centered cubic.
Properties of Diamond:
- Due to the effective sp3 – sp3 overlapping between carbon atoms, diamond possess a high density.
- The force which holds the carbon atoms is very strong and hence it is very difficult to break these bonds. Diamond is such a hard substance that it is used to cut gemstones, glass and for drilling the rocks.
- Diamond has a very high melting and boiling points and has very high refractive index 2.4. It melts at 4000 K. It shows the phenomena of total internal reflection and looks brilliant.
- Since all the four electrons present in the valence shell of carbon are used up in the carbon-carbon sigma bonds and no mobile electrons are available, so it is a non-conductor of electricity.
- Diamond is unstable and is converted to graphite over centuries.






